More Problems
Problem 1:
Find the rotational energy levels of a diatomic molecule with atoms of mass m1 and m2. Take r1 and r2 to be the distances from atoms 1 and 2 to the center of mass.
Solution:
- Concepts:
The rigid rotator - Reasoning:
We model the system as a rigid rotator. - Details of the calculation:
Considering only the rotational motion about the CM, the Hamiltonian of the molecule is L2/(2I) where I is its moment of inertia,
I = m1r12 + m2r22 = μr2, μ = m1m2/(m1 + m2, r = |r1 - r2|.
The eigenfunctions of H are the eigenfunctions of L2.
These are the spherical harmonics, Ylm(θ,φ).
The eigenvalues are El = l(l + 1)ħ2/(2I), l = 1, 2, 3, ... . Each energy eigenvalue is 2l + 1 fold degenerate
The rotational energy levels of the molecule are El = l(l + 1)ħ2/(2I).
Problem 2:
(a) Given that the radial function for the ground state of hydrogen is R(r) = (2/a03/2) exp(-r/a0),
find the value of radius at which the ground state radial probability
reaches a maximum.
(b) What is the probability of finding the ground state electron within the Bohr
radius?
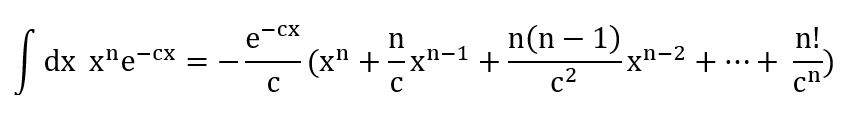
(c) Using your calculation in parts (a) and (b), comment on the similarity and the difference of the hydrogen atom ground state in quantum mechanics compared to the Bohr model of the hydrogen atom (in terms of energy and radius), and give a short explanation of how the quantum mechanical model solves the uncertainty issue in the Bohr model.
Solution:
- Concepts:
The hydrogen atom, fundamental assumptions of QM - Reasoning:
This is a 3-dimensional system.
P(r,θ,φ)d3r = |ψ(r,θ,φ)|2d3r is the probability of finding the electron in an infinitesimal volume d3r about r = (r,θ,φ). ψ(r,θ,φ)| = (4π)-½R(r) for the ground state of the hydrogen atom.
P(r,θ,φ) is the probability per unit volume.
P(r) = ∫02πdφ∫0πsinθ dθ |ψ(r,θ,φ)|2r2 = r2|R(r)|2 = r2 (4/a03) exp(-2r/a0).
P(r) is the probability per unit length. - Details of the calculation:
(a) We are looking for the maximum in P(r).
d(r2exp(-2r/a0))/dr = 2r exp(-2r/a0)(1 - r/a0) = 0 --> P(r)max = P(a0).
The ground state radial probability reaches a maximum at r = a0.
(b) P(r ≤ a0) = ∫0a0P(r) dr = (4/a03) ∫02πdr r2 exp(-2r/a0) = ½∫02πdx x2 exp(-x) = ½(2 - 10e-2).
P(r ≤ a0) = (1 - 5e-2) = 0.323.
(c) Both model calculates the same ground state energy. The Bohr model fixes the radius for the ground that orbital at a0. In QM, the electron does not have a fixed position, but the maximum of the radial probability distribution for the ground state occurs at r = a0.
The Bohr model explicitly violates the Heisenberg Uncertainty Principle, since at any point in the orbit p and r are exactly given. QM resolves this issue by abandoning the idea of a fixed orbit and replacing it with the wave function and the concept of a probability density.
Problem 3:
A particle with mass m is in ground state of 1D harmonic oscillator potential. The
potential energy function is U(x) = ½mω2x2.
(a) A position measurement is made. What is the probability of
finding the particle at the middle of the potential well with a position
uncertainty
Δx << (ħ/(mω))½?
(b) If the particle is found within
Δx at the middle of the potential well, what is the probability that a
subsequent energy measurement will still find the particle in the ground state?
Solution:
- Concepts:
Fundamental assumptions of QM - Reasoning:
Φ0(x) = (mω/(πħ))¼exp(-½mωx2/ħ) is the ground state wave function. Since Δx is very small, the wave function after the measurement is Φ(x) = 1/(Δx)½ within ±Δx/2 of the origin and zero everywhere else.
The probability of finding the particle in this state is |<Φ0|Φ>|2. - Details of the calculation:
(a) |<Φ0|Φ>|2 = |Φ0(0) Δx/(Δx)½|2 = |Φ0(x)|2Δx = Δx (mω/(πħ))½.
(b) Φ(x) is a superposition of energy eigenstates. The probability that a subsequent energy measurement will find the particle in the ground state is|<Φ|Φ0>|2 = Δx (mω/(πħ))½.
Problem 4:
A negative K meson with mass m = 1000 electron masses is captured into a circular
Bohr orbit around a lead nucleus (Z = 82). Assume it starts with principal quantum
number n = 10 and then cascades down through n = 9, 8, 7, ... etc.
(a) What is the energy of the photon emitted in the n = 10 to n = 9 transition?
(b) What is the approximate radius of the lead nucleus if no further quanta are
observed after the n = 4 to n = 3 transition (because of nuclear
absorption of the K meson)?
Solution:
- Concepts:
The hydrogenic atom, the ground-state energy of the hydrogen atom, the Bohr radius - Reasoning:
The lead nucleus and the meson form a hydrogenic atom. The energy levels and the average distance of the meson from the nucleus can be determined from the ground-state binding energy of the hydrogen atom and the Bohr radius. The photon emitted in a transition has an energy equal to the energy difference between the initial and the final state. When the average distance of the meson from the nucleus in the final state becomes of the order of the radius of the nucleus, then a nuclear reaction becomes likely, and the hydrogenic atom will be destroyed. - Details of the calculation:
(a) We have a hydrogen like system with Z = 82.
En = EI'/n2, EI' = EI(μ'/μ)Z2.
μ = 1000 memnucleus/(1000 me + mnucleus) ≈ 1000 me. μ'/μ = 1000. EI = 13.6 eV.
E10 - E9 = 822*13.6*1000* (1/92 - 1/102) eV = 2.2*105 eV.
(b) The radius of the Bohr orbit is rn = n2a0' = n2(a0/Z)(μ/μ') = 9a0/(82*1000) = 5*10-15 m.
This is the approximate radius of the lead nucleus.