

A goal of nuclear physics is to account for the properties of nuclei in terms of mathematical models of their structure and internal motion. A nucleus must be treated quantum-mechanically. We have many interacting quantum-mechanical particles. The Schroedinger equation for a particle in one dimension is
(-ħ2/(2m))∂2ψ(x,t)/∂x2 + U(x)ψ(x,t) = iħ∂ψ(x,t)/∂t.
In 3 dimensions it becomes
(-ħ2/(2m))Ñ2ψ(r,t) + U(r)ψ(r,t) = iħ∂ψ(r,t)/∂t.
where Ñ2ψ(r,t) = ∂2ψ(r,t)/∂x2 + ∂2ψ(r,t)/∂y2 + ∂2ψ(r,t)/∂z2.
For many particles in 3 dimensions we have to write
Σi[(-ħ2/(2mi))Ñi2ψ(r1,r2,r3, ..., t)] + U(r1,r2,r3, ...)ψ(r1,r2,r3, ..., t) = iħ∂ψ(r1,r2,r3, ..., t)/∂t.
This is a many-body
equation that cannot be solved exactly. Approximations are necessary.
In addition, we do not know the nuclear potential
U(r1,r2,r3,
...) very well. It is mainly due to the
strong nuclear force and the repulsive electrostatic Coulomb force. But
nucleons have spins and magnetic moments and since they are very close packed,
magnetic interactions also become important.
Two approximations are often the starting point for calculation. They are then refined by adding extra terms as small perturbations.
So there are two basic types of simple nuclear models.
The Shell Model (developed by Maria Goeppert-Mayer and Hans Jensen), emphasizes the orbits of individual nucleons in the nucleus. The Nuclear Shell Model is similar to the atomic model where electrons arrange themselves into shells around the nucleus. The atomic shell structure is due to the quantum nature of electrons and the fact that electrons are fermions. Since protons and neutrons are also fermions, the energy states of the nucleons are filled from the lowest to the highest as nucleons are added to the nucleus. In the shell model the nucleons fill each energy state with nucleons in orbitals with definite angular momentum. There are separate energy levels for protons and neutrons.
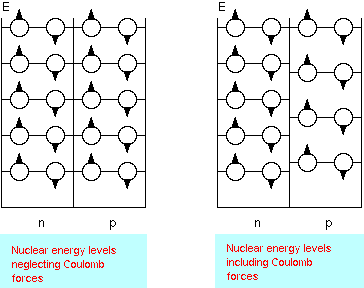
The ground state of a nucleus has each of its protons and neutrons in the lowest possible energy level. Excited states of the nucleus are then described as promotions of nucleons to higher energy levels. This model has been very successful in explaining the basic nuclear properties. As is the case with atoms, many nuclear properties (angular momentum, magnetic moment, shape, etc.) are dominated by the last filled or unfilled valence level.
The nuclear shell model explains the existence of "magic numbers". Nuclei with magic neutron number N = 2, 8, 20, 28, 50, 82, 126 or magic proton number Z = 2, 8, 20, 28, 50, 82 have a larger binding energy per nucleon than neighboring nuclei, and when N and Z are both magic the binding energy per nucleon is especially large. This suggests a shell structure, similar to the shell structure in atomic physics, where the noble gases have especially large ionization energies.
What is the origin of this shell structure? Consider a single nucleon in a nucleus. All the nucleons form a nuclear "fluid". Our single nucleon interacts with all the other nucleons, but the interactions change quickly and only treating the attraction of all the other nucleons as a function of position is a good approximation. We postulate an average nuclear potential as shown in the figure below.
![\includegraphics [width=8cm]{AverageField}](images/potnuc.gif)
The 3-dimensional spherical well or the isotropic three-dimensional harmonic oscillator potential allow analytical solutions. Let us consider the three-dimensional harmonic oscillator potential. The time-independent Schroedinger equation for this system is
(-ħ2/(2m))Ñ2ψ(r,t) + (1/2)mω2r2ψ(r,t) = Eψ(r,t),
Seperation of variables is possible in Cartesian as well as in spherical coolrdinates. Expressed in spherical coordinates, the eigenfunctions and eigenvalues of this system are
ψ(r,t) = Rnl(r)Ylm(θ,ϕ), Enl = (2n + l + 3/2)ħω,
with n = 0, 1, 2, 3, ..., and
l
= 0, 1, 2, ... .
Using Cartesian coordinates we write the eigenvalues as
Enx, ny, nz = Enl = (nx + ny + nz + 3/2)ħω, nx, ny, nz = 0, 1, 2, ... .
This simple model generates the first three magic numbers.

The model fails for the larger magic numbers because it
neglects the spin-orbit energy, which splits the levels by an amount which
increases with orbital quantum number.
B(Z,N) = C1A - C2A2/3 - C3Z(Z-1)/A1/3 - C4(N-Z)2/A.
The liquid drop model of the nucleus takes into account that the
forces on the nucleons on the surface are different from those on
nucleons in the interior, where they are completely surrounded by other
attracting nucleons. This is similar to considering the surface
tension to be a contributor to the energy of a tiny liquid drop.
The first term of the semi-empirical formula is the volume term.
It is proportional to the mass number A. If we divide by A, this
term alone would gives a constant prediction for B(Z,N)/A, which for
large nuclei is nearly, but not completely, correct.
The second term in the formula is the surface term, which is
proportional to A2/3. It decreases the binding energy
because nucleons near the surface of the nucleus are in a less deep
potential well. The surface term is most important for the binding
energy of small nuclei, because the ratio of the second to the first
term is largest for small A.
The third term of the semi-empirical formula is the Coulomb term.
Each pair of protons contributes to the Coulomb term equally. The
number of protons is Z, so the number of proton pairs is Z(Z-1)/2.
The electrostatic potential energy is proportional to the inverse
distance. The average inverse distance increases with the radius
of the nucleus and therefore is proportional to A1/3.
The total electrostatic potential energy term is proportional to
Z(Z-1)/A1/3, and it deceases the binding energy.
The fourth term of the formula expresses the fact that the binding
energy decreases if a neutron is transformed into a proton, if the
number of protons is larger than that of the neutrons. The same
holds if a proton is transformed into a neutron.
With increasing Z the magnitude of the Coulomb term increases at the highest rate. For some value of Z the magnitude of the Coulomb term will become larger than the volume term and the nucleus will become completely unstable. No nuclei with Z > 120 can exist. This is a direct consequence of the long range nature of the Coulomb force.
The Shell Model and the Collective Model represent the two extremes of the behavior of nucleons in the nucleus. More realistic models, known as unified models, attempt to include both shell and collective behaviors.